For Medical Device Industry
MaRVis MR devices are essential tools for innovative, powerful, and patient-friendly minimally invasive medical procedures guided by magnetic resonance imaging (MRI).
Technology
Commercially available guidewires for X-ray-guided interventions are built from metal wires. In MRI the metal parts heat up and conduct electricity. This leads to injury to the patient and the physician. Therefore, conventional guidewires are MR unsafe and must not be used in MRI. The same applies to metal-braided or -coiled catheters.
The MaRVis platform technology integrates good material properties and concomittant elimination of their critical electrical features in MRI by using non-metallic fiber-composite materials. This proprietary technology by de novo design of medical devices obviates the use of metal materials and provides for a high degree of flexibility in their mechanical and visualization properties.
Monobloc design
Micro guidewires consist of one MaRVis rod. 0.035” guidewires consist of several MaRVis rods which are geometrically arranged. Stiffness of the 0.035” guidewires is modulated by incorporating three peripheral (standard version) or six peripheral (stiff version) MaRVis rods. Geometric arrangement of the MaRVis rods is achieved by embedding in an envelope polymer.
The MaRVis rods extend along the entire length of a guidewire so that a monoblock guidewire design is realized and similarly can be realized for other MR devices.

MaRVis rods
The proprietary basic material for the design of MaRVis MR Guidewires is a thin rod-shaped composite material (“MaRVis rod”). These rods are composed of glass or aramid fibers, or in certain designs of both fibers together, impregnated by epoxy resin. In appropriate way these MaRVis rods can also be used for other medical devices, e.g. MR catheters.
Product performance and safety
The glass fibers in the MaRVis rods contribute to the mechanical strength of the guidewires. A MaRVis MR Guidewire provides high stiffness (adapted to the respective type of guidewire) and transmits compressive and tractive forces as well as torsional moment. This design allows handling according to today’s established standards. All MaRVis Guidewires contain aramid fibers and a PTFE heat shrink tube, both providing high elasticity and tear-resistance, and thereby product safety. The smooth PTFE surface ensures good gliding properties.
MaRVis MR marker system
MaRVis has developed an elaborated MR marker system based on iron microparticles which function as passive-negative markers in MRI.
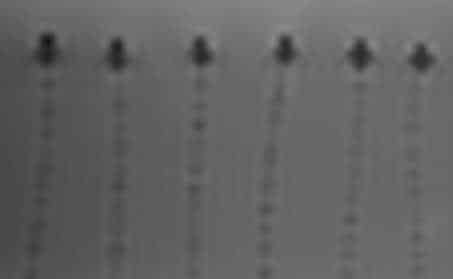

MaRVis platform technology
The patent-protected platform technology allows development of MR safe and visible guidewires, mandrels, catheters and other medical devices, based on the same basic design principles as described above for the MaRVis MR Guidewires. All devices will benefit from the good mechanical properties achievable and the continuous line MR visibility plus the discrete MR tip marker for identification of the very distal end of the device.
Contact
For more information about MaRVis Interventional, it´s products, technology and cooperation opportunities please contact us:
